|
The carboxyl group consists of a carbon, bonded to both an oxygen and a hydroxyl group. The carboxyl group is seen in many organic molecules known as carboxylic acids, which have a variety of functions. In general, carboxylic acids are represented by the formula RCOOH, where R is a hydrocarbon group. A carboxyl group is one of many functional groups that attaches to larger molecules and gives them certain properties. Carboxylic Acid Functional Group Carboxylic acids are organic compounds (hydrocarbons) that contain the element oxygen. A carboxyl group is a carbonyl group (C O) with a hydroxyl group (-OH) attached to the. A carboxyl group consists of a carbon double-bonded to oxygen and singly bonded to a hydroxyl group. The functional group in carboxylic acids is called the carboxyl group. In this structure of a carboxyl group, a carbon atom is attached to an oxygen atom with the help of a double. It can be referred to as a carboxy group, a carboxyl functional group, or a carboxyl radical. Carboxyl group is a functional organic compound. As with aldehydes and ketones, carboxylic acid formulas can be written to show the carbon-to-oxygen double bond explicitly, or the carboxyl group can be written in condensed form on one line. A carboxyl is a common functional group in organic chemistry. The adsorption mechanism of the removal of lead from water by using carboxylic functional group (COOH) functionalized on the surface of carbon nanotubes was. The carboxyl group is a functional group that contains a carbon–oxygen double bond and an OH group also attached to the same carbon atom, but it has characteristic properties of its own. Carboxylic acids are compound containing carboxyl structure. It also has a single bond to a hydroxyl group. In this structure of a carboxyl group, a carbon atom is attached to an oxygen atom with the help of a double bond. is an organic compound that has a carboxyl group A functional group that contains a carbon–oxygen bond and an OH group also attached to the same carbon atom. functional group name polar/nonpolar reactivity/notable trait. Carboxyl group is a functional organic compound. However, in these compounds, the carbonyl group is only part of the functional group.Ī carboxylic acid An organic compound that has a carboxyl group. 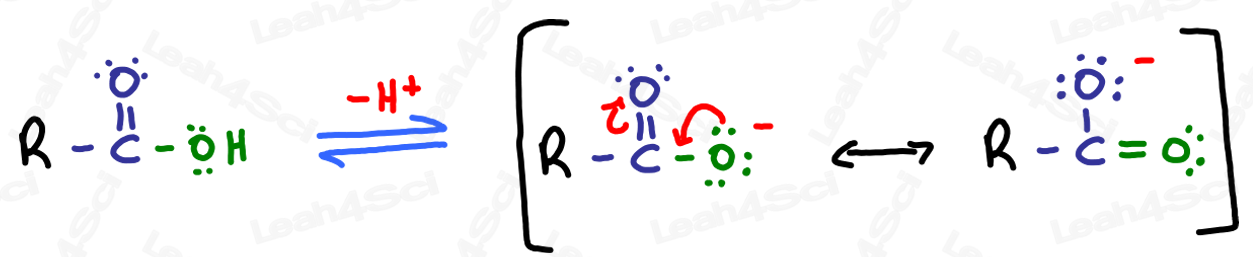
The carbonyl group is also found in carboxylic acids, esters, and amides. We introduced the carbonyl group (C=O)-the functional group of aldehydes and ketones-in Chapter 14 "Organic Compounds of Oxygen". Identify the functional group for a carboxylic acid, an ester, an amine, and an amide.An indispensible resource for the organic chemist. Identify the general structure for a carboxylic acid, an ester, an amine, and an amide. Patais Chemistry of Functional Groups is one of chemistrys landmark book series in organic chemistry.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
